
If medical technology products get you excited and make your heart beat faster, MT-CONNECT is the perfect place for you. You’ll find all the industry’s innovations under one roof here – and that’s not the only reason to pay the exhibition a visit…
We cordially invite you to the MT-Connect in Nuremberg and look forward to your visit.
Your free admission ticket is available here:
[contact-form-7 404 "Not Found"]We are pleased to announce that we will be exhibiting at MT-Connect on 21 – 22 June in Nuremberg, Germany. We welcome you to visit our stand Hall 10.0 / 10.0-214 to discuss our new product lines or tell us any other ways we may be able to help your business.
We present the ACSYS MEDICAL program, which is subdivided into three components: the ACSYS MEDICAL process validation and device qualification, the ACSYS MEDICAL complete laser process and the ACSYS MEDICAL laser processing systems specially adapted to medical technology.
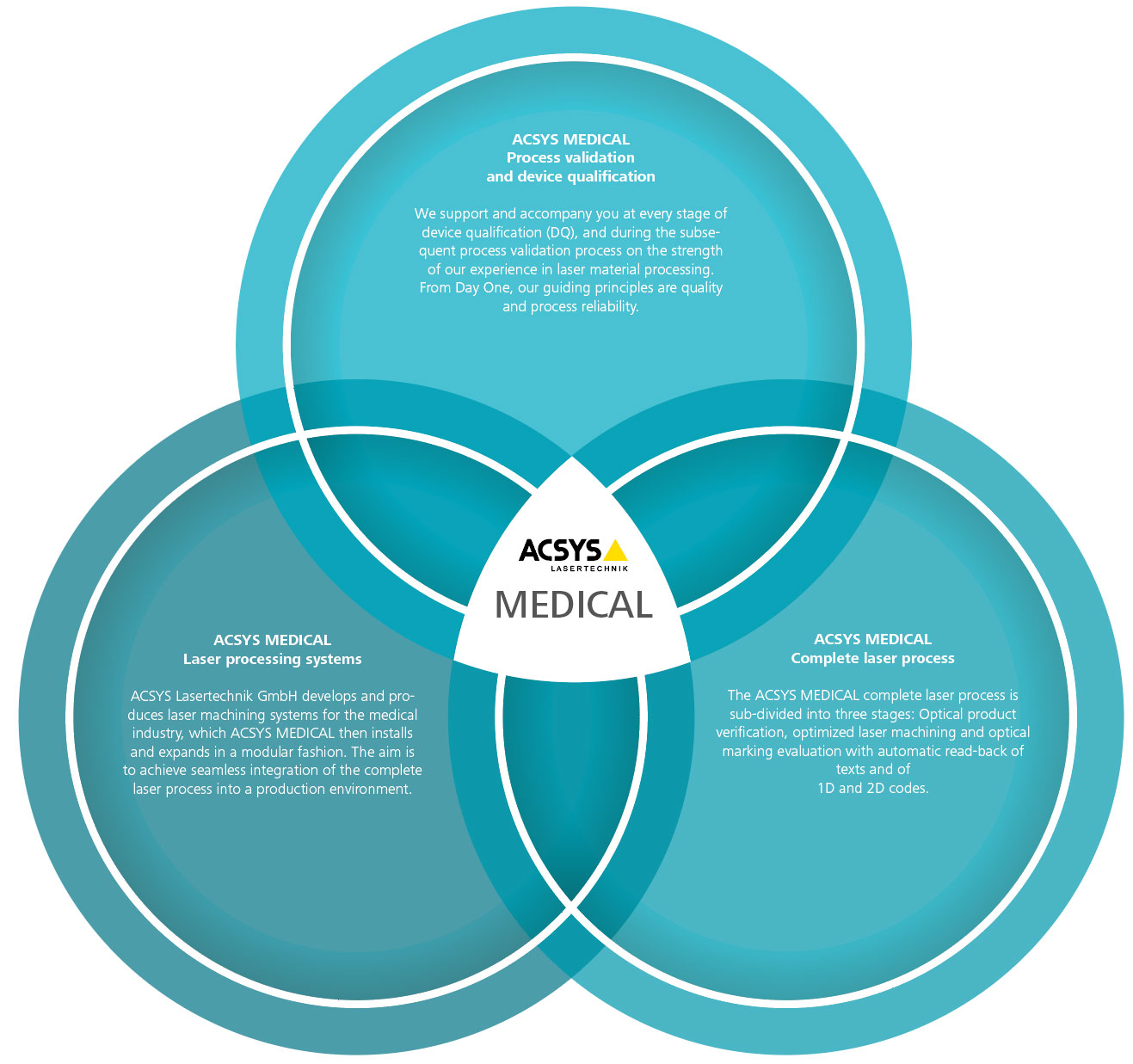
ACSYS MEDICAL Process validation
and qualification of laser system solutions
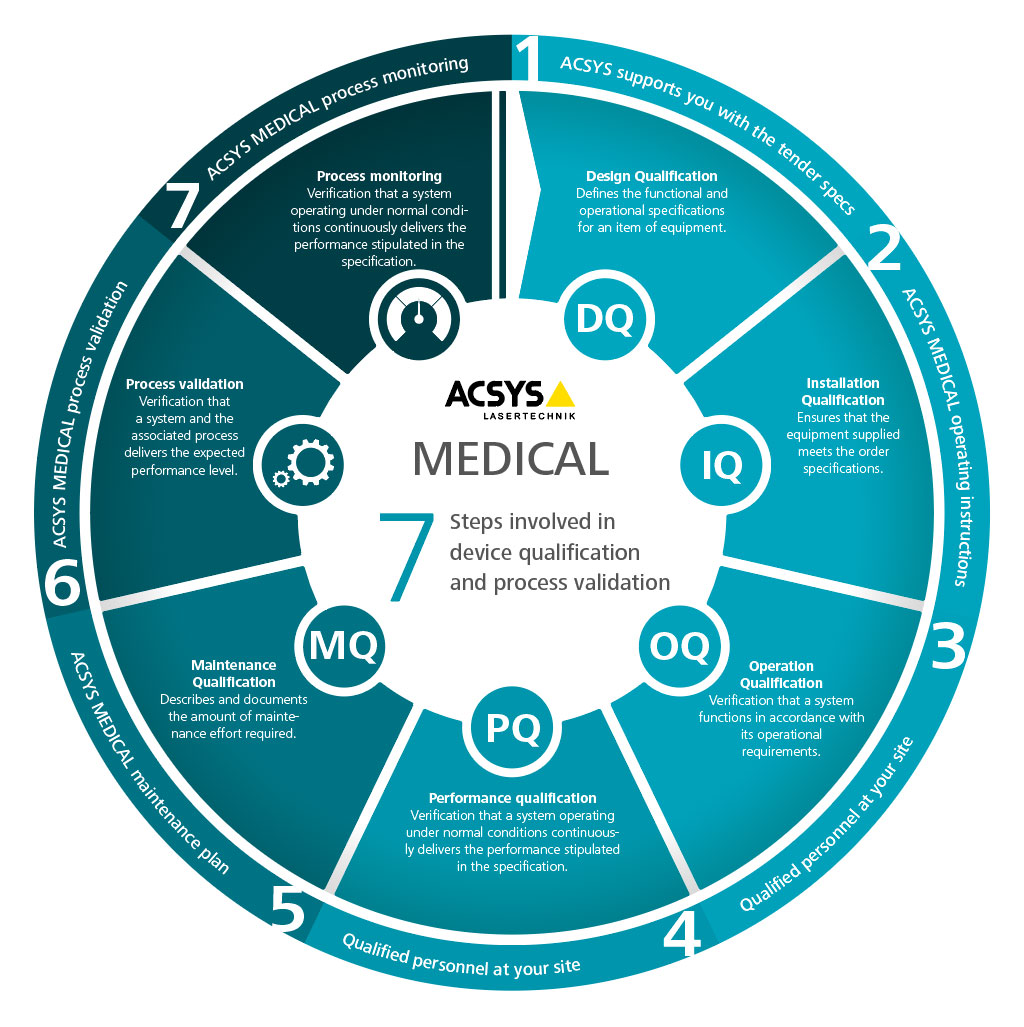
The qualification of laser machining systems for the manufacture of medical products is a clearly defined requirement from the “Rules of Good Manufacturing Practice – GMP”, and is therefore an indispensable element of the quality assurance process. This guide provides an overview of the specifics governing how we should proceed with the qualification process – in particular with respect to the inclusion of individual activities in the planning and building of complex laser systems.
Right from the outset, ACSYS is there to provide you with detailed technical knowledge in relation to the FDA-compliant qualification (US Food and Drug Administration) of laser units in the medical industry.
ACSYS MEDICAL laser process in medical technology
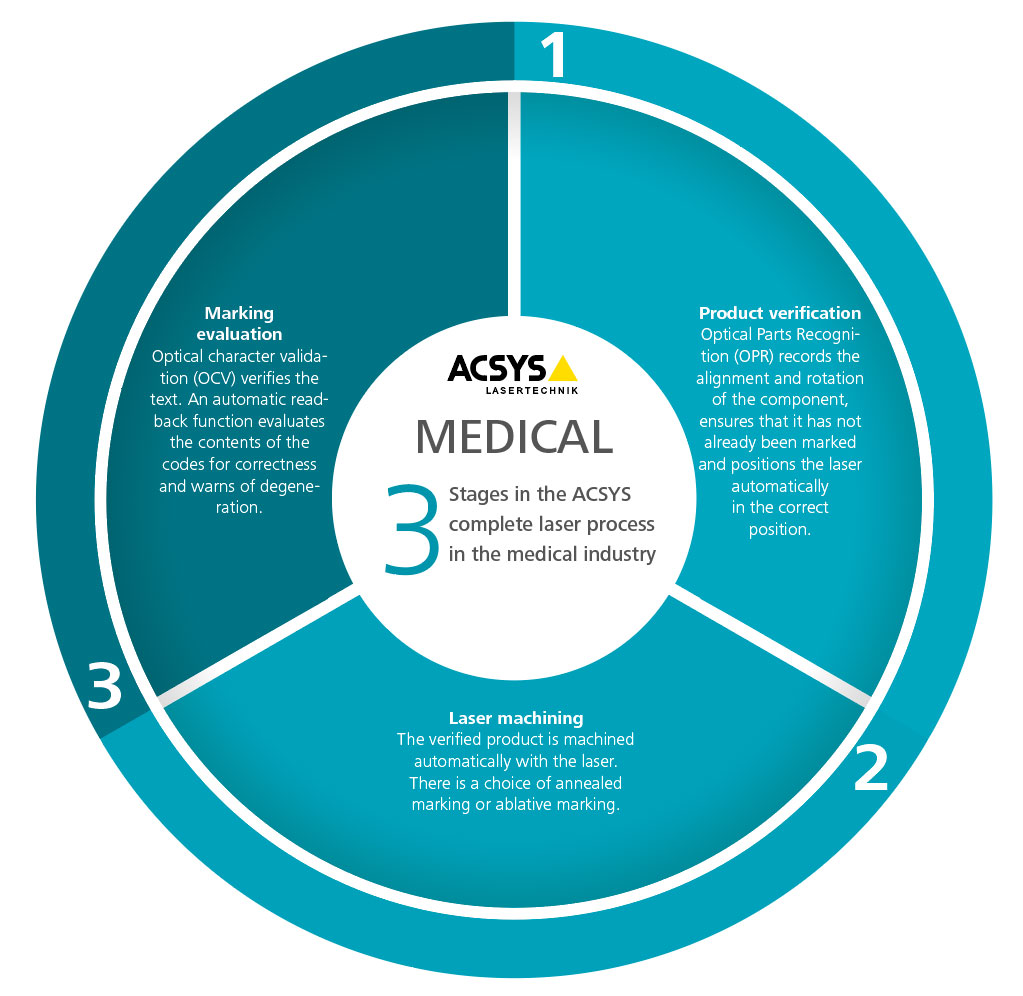
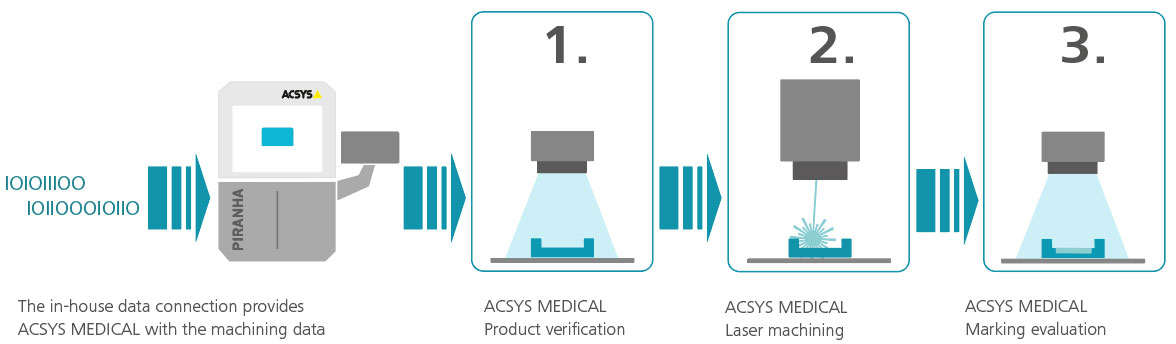
Laser marking and laser engraving are just one part of the overall process that ACSYS can display in medical technology. Our holistic approach and close collaboration with customers enables ACSYS to offer modular and customer-specific solutions. Our in-house software development department produces the appropriate software with database connections, component recognition, the corresponding protocol functions and concluding read-back functions of barcodes and DataMatrix codes as well as plain text entries.
1. Product verification
2. Laser machining
3. Marking evaluation
ACSYS laser systems are manufactured in accordance with GMP recommendations. A high level of vertically integrated production means that all necessary hardware requirements can be taken into account, required for subsequent operation in the manufacture of medical products.
ACSYS MEDICAL Laser processing systems
The portfolio of solutions from ACSYS provides the evidence: Through the application of new technological solutions, no boundaries are set in stone for ever. We have been enhancing our laser machining system with automation and robotics for some considerable time already. For this, we use the skills of our team of specialist development engineers and ultra-modern 3D design tools and many years of expertise. This enables us to can bring to bear a unique technological creative potential on task profiles, and to do so in a way that no-one has previously dared to venture.
- Robotics
- Automation
- Multiple laser and multi-head systems
- Conveyor belt and roller supply
- Palletizing and Pick & Place
- Rotary indexing table and cross table
- Film and rating plate handling
At the MT-Connect, ACSYS shows the new PIRANHA II Multi.

This machine at the top of the ACSYS product range offers high performance and quality with a minimum footprint. Fiber laser systems with exceptional beam quality make the PIRANHA a unique production machine for laser marking and engraving.
The new developments do justice to the ACSYS philosophy of placing customer benefit at the core of their actions. The modular design of the laser systems allows customer-specific configuration and thus also remains open for future hardware and software modifications and extensions.

Messe Nuremberg, Hall 10.0 / 10.0-214.
Challenge us!
We work “live” on your workpieces and most different materials!
- Date: 21 – 22 June 2017
- Venue: Messe Nuremberg
- Opening times: Daily 10.00 a.m. to 6.00 p.m.
Do you need more information, have questions for Transportation Travel and overnight accommodations?
Call us or write to us.
Tel.: +49 7154 808 75 0
Fax: +49 7154 808 75 19
E-Mail: messe@acsys.de
